The Particle Model Of Light MDCAT Quiz with Answers
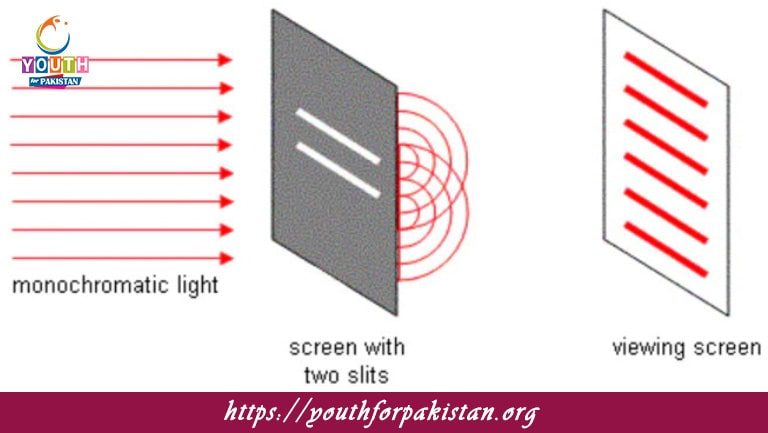
The Particle Model Of Light MDCAT Quiz is a theory that describes light as being composed of discrete packets of energy called photons. That is, the model was developed to account for phenomena like the photoelectric effect, where light striking a metal surface ejects electrons from the surface. While the particle theory was first propounded by Isaac Newton, it was significantly further refined by Albert Einstein at the beginning of the 20th century. Einstein’s explanation of the photoelectric effect—work for which he was awarded the Nobel Prize—demonstrated that light has properties consistent not only with a wave (as previously thought) but also with a stream of particles, or photons. In fact, this particle-like behavior of light is fundamental in several quantum phenomena. For MDCAT students, the most vital part of studying the particle model of light is to handle questions on the nature of light and its interaction with matter; more importantly, the foundation of quantum mechanics.
Test Your Knowledge with an MDCAT Quiz
An MDCAT Quiz on the Particle Model of Light is designed to strengthen your learning of how light sometimes acts as a particle and sometimes as a wave. It will, therefore, include the photoelectric effect, the concept of photons, and how light transforms from a classical view to the quantum view. By doing these quizzes, you would be in a better position to answer any question confidently relating to the particle nature of light and its applications in modern Physics, particularly in the MDCAT exam.
- Test Name: The Particle Model Of Light MDCAT Quiz
- Type: Quiz Test
- Total Questions: 30
- Total Marks: 30
- Time: 30 minutes
Note: Answer of the questions will change randomly each time you start the test, once you are finished, click the View Results button.
Free Flashcards for Quick Revision
Free Flashcards on the Particle Model of Light summarize important concepts concisely, such as photon energy, the photoelectric effect, and the historical development of light theory. These flashcards are a great revision tool that will help you quickly recall important definitions and formulas, ensuring you are well-prepared for any light-related questions in the MDCAT exam.

The photoelectric effect provided evidence for the particle nature of light by demonstrating that:
Light can eject electrons from a metal

In the photoelectric effect, increasing the intensity of light:
Does not affect the energy of the ejected electrons

According to Einstein's explanation of the photoelectric effect, electrons are ejected from a material when:
They absorb energy from photons

The discovery that light can be thought of as both a wave and a particle is referred to as:
Wave-particle duality
Experience the real exam environment with our expertly designed collection of over 25,000 MCQs MDCAT Mock Tests.